


From a materials science perspective, the solutions of biology are outstanding, despite a limited selection of substances determined by the availability of chemical elements. This is achieved not least by the fact that different units work together synergistically by interlocking them hierarchically on different length scales. In research one is not subjected to the restrictions of availability and has access to the entire range of functional bodies. In the chemical materials sciences, however, it has so far only been possible to a limited extent to create cooperative effects in a similar way through hierarchical structuring. This motivates us, and we are developing Molecular Mesotechnology as a tool to achieve this goal.
Molecular Mesotechnology
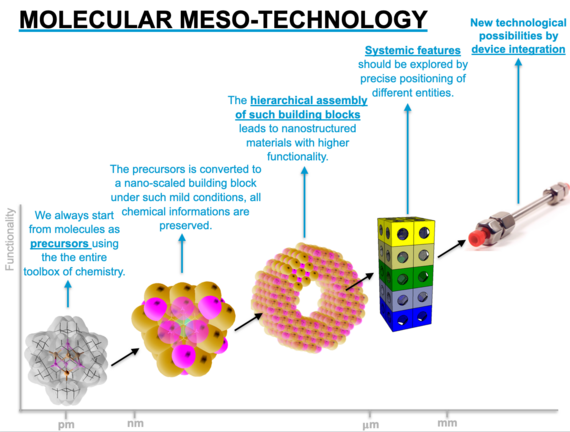

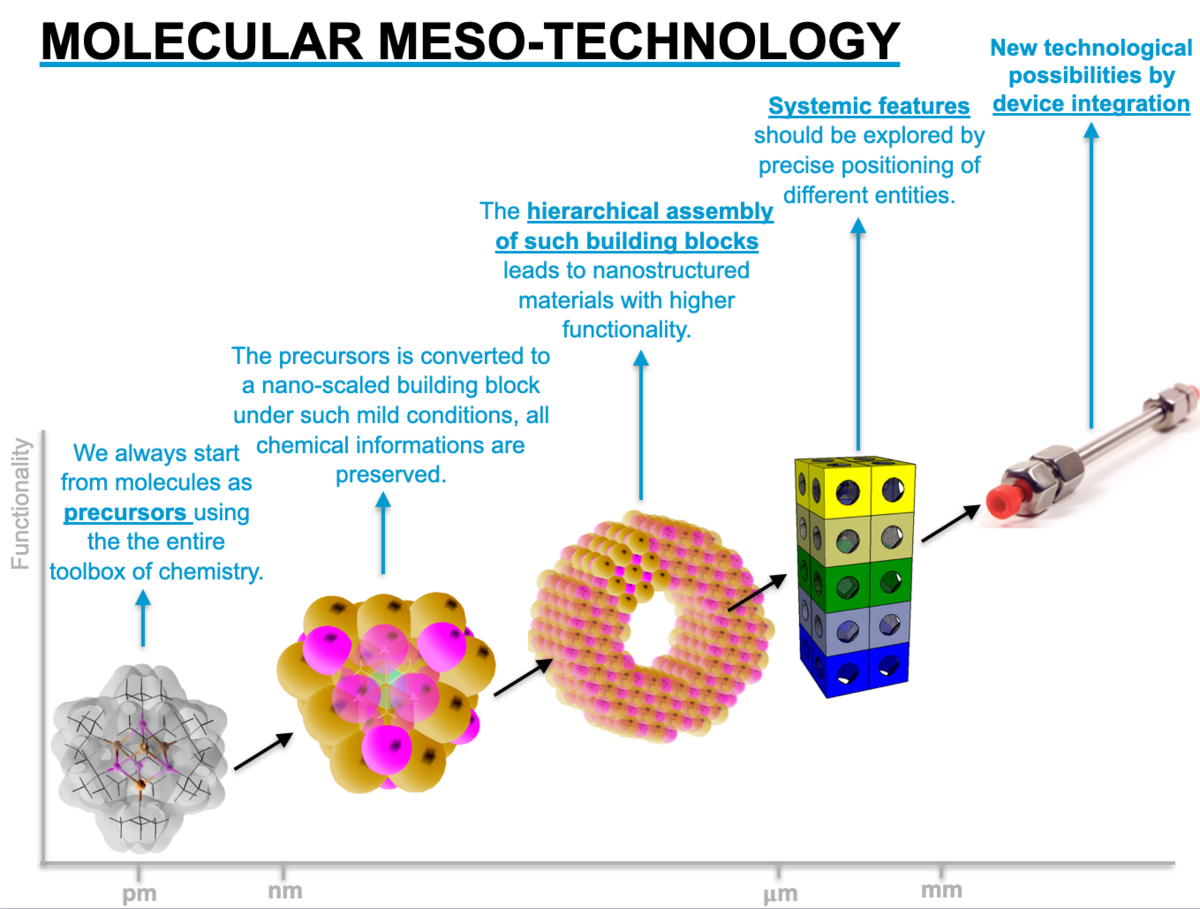
We use the entire arsenal of chemical synthesis (inorganic, organic, organometallic) to create molecular assemblies in which certain functional properties are stored. These can be organic groups in organosilicate sol-gel precursors, surfactants with metal-containing heads or also precursors for nanoparticle syntheses. The information is retained during the mesostructuring of the solids starting from these precursors, so that a higher degree of control over the functional properties can be achieved. In order to understand the properties in detail, an equally broad basis of material analysis is required. Finally, the materials obtained are used as models for a concrete application.
Self-Assembly: Surfactants with novel head groups
Surfactants are molecular representatives of so-called amphiles, substances that have both a water-compatible (hydrophilic) part and an oil-compatible (hydrophobic) part. Surfactants are characterised by a variety of self-assembly properties. The best known of these is the formation of micelles. They have significance in a large number of applications, e.g. in cleaning agents, cosmetics, pharmaceuticals, etc. The AG Polarz is specialized in the synthesis of surfactants with a metal-containing head group to create novel and highly functional systems.
A major focus is on surfactants that are characterized by catalytic properties. Specifically, we deal with surfactants that have head groups:
- Fullerenes as head group for photocatalysis
- Metal N-heterocyclic carbene complexes as head groups for cross-coupling
We are also fascinated to investigate how the self-organisation behaviour of surfactants changes in a non-equilibrium situation. For this purpose, surfactants must be available into which energy can be introduced from external sources. That is why we develop stimulus-responsive surfactant systems. Specifically, we deal with surfactants that have head groups:
- Molecular semiconductors and their redox activity
- Molecular semiconductors and their redox activity
Semiconductor materials: particle-based materials



In particle-based materials, the property profile of the individual nanoparticle is supplemented with additional characteristics by the collective behaviour of the ensemble. Since the spatial arrangement of the particles should therefore not be random, the role of the particle shape becomes extremely important. A periodic arrangement of single objects in space increases the chances to observe synergistic effects independent of scale, i.e. characteristics that only develop for an ensemble. On the atomic scale, an example is the formation of band structures caused by the electronic interaction of atoms at lattice positions.
Particles larger than atoms can also form crystalline phases, in the simplest case the densest sphere packings. The so-called photonic materials are an impressive example of the fact that periodic assembly results in collective effects. In contrast to atoms, however, in the case of particles there can also be non-spherical structural units, which opens up other possibilities of arrangement. Which cooperative properties will result for these special colloidal crystals is a current question in the field of particle-based materials, the answer to which is inseparably linked to a number of scientific problems. First the properties of the individual particle must be considered. It has long been known that for the same composition, the properties are very strongly influenced by the precise adjustment of the expansion of a solid. The underlying size quantification effects represent the elementary basis of the nanosciences. The question to be answered is whether, with the same volume, the shape of the particle also has an effect on its properties. Ultimately, the starting point of the analysis is that different, or even better, arbitrary particle shapes can be generated for a desired functional material.
Our key interests:
- Nanoparticles from magnetic semiconductors e.g. EuO
- Organic-inorganic perovskite materials for optoelectronic applications
- Zinc oxide materials and form dependent self-assembly
- Photonic glasses
- Oxidic barcode-nanowire as memristors
Porous solids: gradient materials and neighbouring group effects
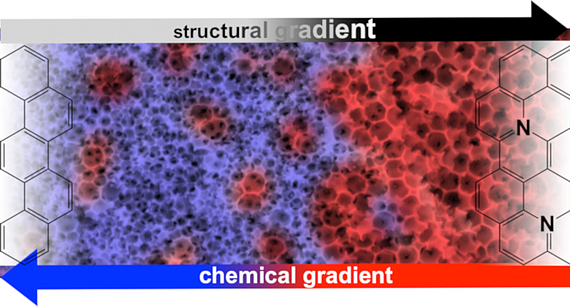
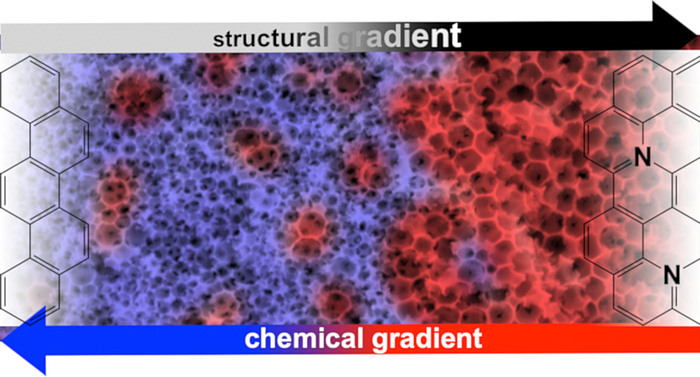
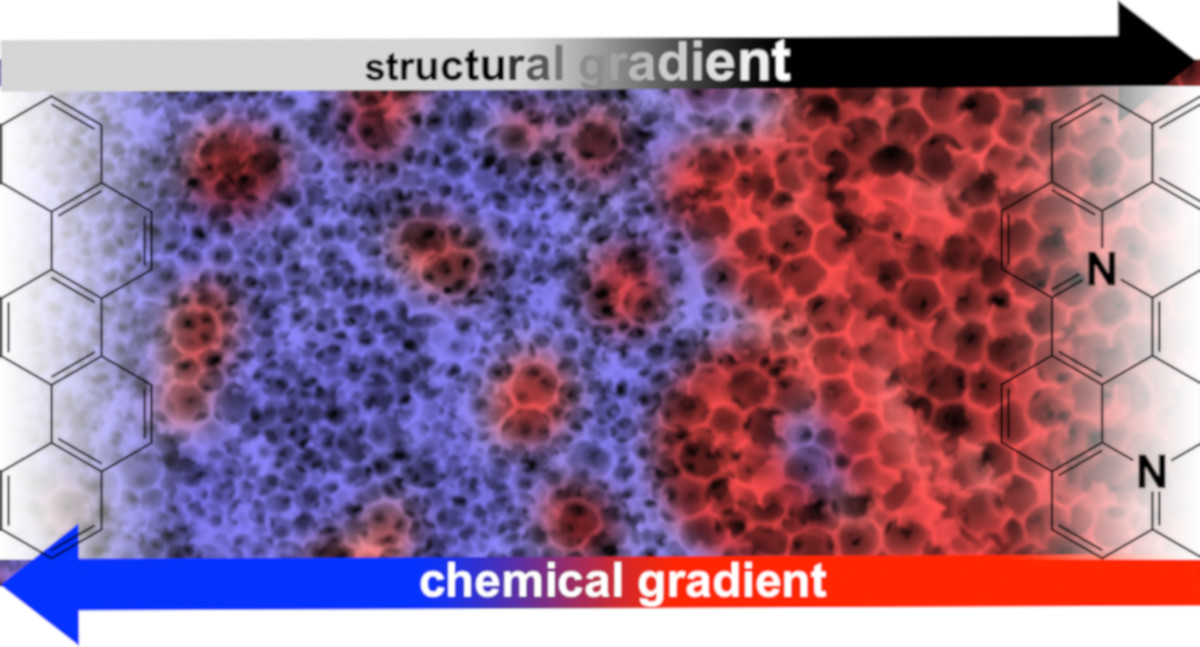
The homogeneity of a material is generally regarded as a criterion of quality, which is why the syntheses are usually carried out in such a way that there are no local differences in structure and composition. On the other hand, there are so-called functional gradient materials, which can perform much more complex tasks by moving from one property to another. We try to transfer this concept to the field of porous materials. Ultimately, this also involves the question of how different functional groups in a material can interact in such a way as to produce new, cooperative effects and properties.
Specifically, we deal with:
- Porous Janus-type nanoparticles as active colloids
- Porous gradient materials through click-chemistry
- Porous organosilicate materials for biocidal coatings and as living functional materials Chemistry
- Neighbouring group effects in chromatography and for CO2 storage materials
- Porous carbon materials with functional gradients
Cooperations
Head of the group


30167 Hannover














